This Student Recreated the Universe in a Bottle. What She Discovered Could Help Reveal How Life Started on Earth
A study reconstructs the chemical composition of key molecules in stars by analyzing their infrared fingerprints.
A PhD student has recreated a small portion of the Universe inside a laboratory setup, producing synthetic cosmic dust and offering new insight into how the chemical ingredients for life may have formed long before Earth existed.
Linda Losurdo, a PhD candidate in materials and plasma physics at the University of Sydney’s School of Physics, conducted the experiment using a simple blend of gases—nitrogen, carbon dioxide, and acetylene. The setup was designed to replicate the extreme and highly dynamic conditions found around stars and within supernova remnants.
When exposed to strong electrical energy, the gas mixture produced carbon-rich particles that resemble cosmic dust found drifting through interstellar space and within comets, asteroids, and meteorites.
The findings have been published in The Astrophysical Journal of the American Astronomical Society.

The lab-created material contains a complex combination of carbon, hydrogen, oxygen, and nitrogen, commonly referred to as CHON molecules. These elements form the basis of many organic compounds that are essential for life.
“We no longer have to wait for an asteroid or comet to come to Earth to understand their histories,” Ms Losurdo said. “You can build analog environments in the laboratory and reverse engineer their structure using the infrared fingerprints.
“This can give us huge insight into how ‘carbonaceous cosmic dust’ can form in the plasma puffed out by giant, old stars or in cosmic nurseries where stars are being born and distribute these fascinating molecules that could be vital for life.
“It’s like we have recreated a little bit of the Universe in a bottle in our lab.”

Infrared signatures validate cosmic analog
Cosmic dust forms in highly energetic regions of space, where molecules are continuously struck by charged particles such as ions and electrons. Scientists detect this material by measuring its infrared emission, which acts as a molecular fingerprint revealing its composition.
The dust produced in the laboratory displayed the same distinctive infrared patterns, showing that the experiment closely reproduces the processes that occur in space.

Extraterrestrial material may seed life
One of the major questions in science is how life first began on Earth. Researchers continue to explore whether early organic molecules formed on the planet itself, arrived later via comets and meteorites, or were delivered during the earliest stages of solar system formation, or some combination of these possibilities.
Between about 3.5 and 4.56 billion years ago, Earth experienced heavy bombardment from meteorites, micrometeorites, and interplanetary dust originating from asteroids and comets. These impacts likely delivered large amounts of organic material to the planet’s surface, though the origin of that material is still uncertain.

“Covalently bonded carbon and hydrogen in comet and asteroid material are believed to have formed in the outer envelopes of stars, in high-energy events like supernovae, and in interstellar environments,” Ms. Losurdo said.
“What we’re trying to understand are the specific chemical pathways and conditions that incorporate all of the CHON elements into the complex organic structures we see in cosmic dust and meteorites.”
Laboratory simulation recreates cosmic chemistry
In the experiment, Ms. Losurdo and her supervisor, Professor David McKenzie, used a vacuum pump to remove air from glass tubes, recreating the near-empty conditions of space. Nitrogen, carbon dioxide, and acetylene were then introduced into the system. The gas mixture was exposed to an electrical potential of around 10,000 volts for about an hour, forming a plasma known as a glow discharge.
Under these high-energy conditions, molecules broke apart and recombined into more complex structures. These compounds eventually settled as a thin layer of dust on silicon chips placed inside the tubes.
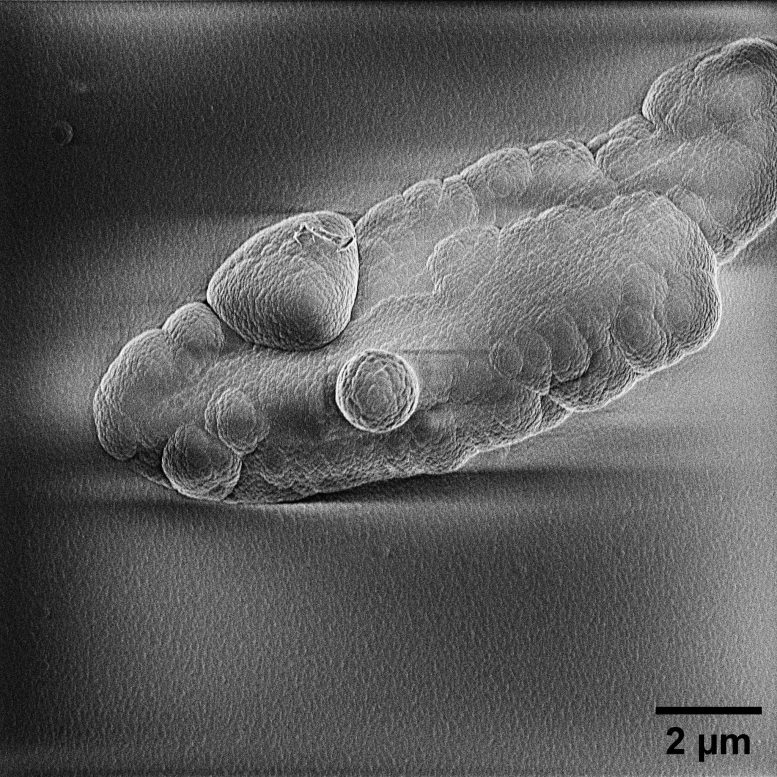
The resulting material sometimes appears as sparkling, glitter-like particles.
Professor David McKenzie, co-author on the paper, said the approach allows scientists to investigate environments that cannot be directly observed in space.
“By making cosmic dust in the lab, we can explore the intensity of ion impacts and temperatures involved when dust forms in space,” Professor McKenzie said. “That’s important if you want to understand the environments inside cosmic dust clouds, where life-relevant chemistry is thought to be happening.
“This also helps us interpret what a meteorite or asteroid fragment has been through over its lifetime. Its chemical signature holds a record of its journey, and experiments like this help us learn how to read that record.”
Spectral database will guide observations
In addition to studying the origins of life, the researchers aim to build a detailed database of infrared signatures from laboratory-produced cosmic dust. This resource could help astronomers identify promising regions of space, such as stellar nurseries or the remnants of dead stars, and better understand the processes shaping them.
By recreating cosmic chemistry under controlled conditions, the work provides a new way to study how complex molecules form in space and offers insight into the early processes that may have contributed to the emergence of life on Earth.
Source: SciTechDaily
This Metal Melts in Your Hand – and Scientists Just Discovered Something Strange
This Student Recreated the Universe in a Bottle. What She Discovered Could Help Reveal How Life Started on Earth/This Student Recreated the Universe in a Bottle. What She Discovered Could Help Reveal How Life Started on Earth
