The transcription factor Lef1 switches partners from β-catenin to Smad3 during muscle stem cell quiescence
Muscle stem cells swap coactivators
After birth, skeletal muscle stem cells (MuSCs) transition from a proliferative to a quiescent state.
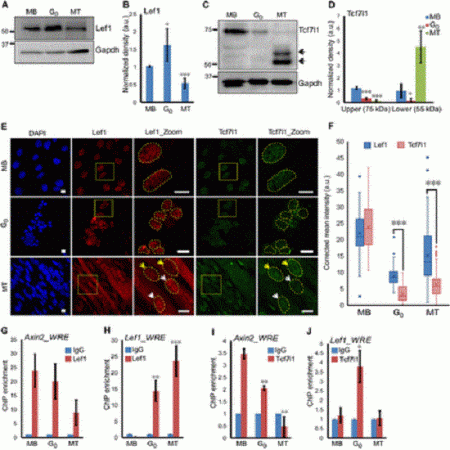
These quiescent stem cells, also known as satellite cells, retain the ability to self-renew and, upon activation, re-enter the cell cycle to generate myoblasts for muscle repair. The entry of MuSCs into quiescence is characterized by the expression of target genes that are activated by the Tcf and Lef (Tcf/Lef) family of transcription factors. Aloysius et al. found that, although Tcf/Lef transcription factors promoted gene expression by cooperating with β-catenin in response to Wnt signaling in proliferating myoblasts, β-catenin was not required for the induction of Tcf/Lef target genes during quiescence. Instead, Lef1 partnered with the transcriptional coactivator Smad3. Smad3 mediates transcriptional responses to transforming growth factor–β (TGF-β) signaling, implicating cross-talk between the Wnt and TGF-β pathways in controlling MuSC quiescence.