Artificial ovary? First atlas of human ovary, a fertility breakthrough
The first “atlas” of the human ovary could lead to restoring ovarian hormone production and the ability to have biologically related children.
Researchers from the University of Michigan (U-M), part of the Human Cell Atlas project aiming to map all the different cells, have broken significant ground.
They have developed a potential means to create artificial ovaries using tissues that were stored and frozen before exposure to chemotherapy and radiation.
Artificial ovaries
At present, surgeons can implant previously frozen ovarian tissue to restore hormone and egg production, albeit temporarily.
But this does not work for too long. According to the researchers, few follicles that produce hormones and carry eggs survive through reimplantation.
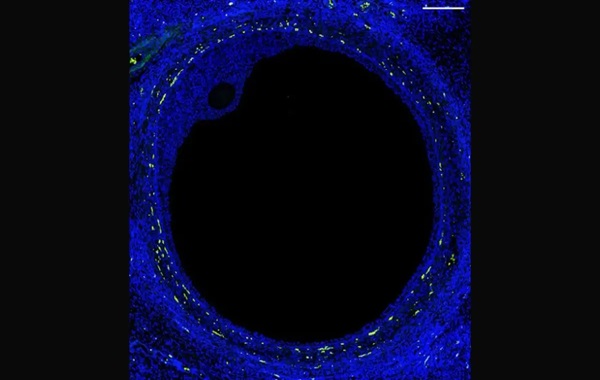
The new “atlas” is particularly significant as it discovered the factors that help a follicle to mature. Most follicles fade away without releasing hormones or an egg.
With new tools that identify what genes are being expressed at a single-cell level within a tissue, they were able to zero in on ovarian follicles that carry the immature precursors of eggs. These are known as oocytes.
Many conditions can prevent eggs from maturing and being released. These include genetic factors, hormonal imbalance, and exposure to certain environmental toxins and pollutants. Even some medical treatments, such as chemotherapy or radiation therapy, can damage ovarian tissue and affect the maturation of oocytes.
The team leveraged spatial transcriptomics to track all gene activity in tissue samples. This new technology reads strands of RNA.
“Now that we know which genes are expressed in the oocytes, we can test whether affecting these genes could result in creating a functional follicle. This can be used to create an artificial ovary that could eventually be transplanted back into the body,” said Ariella Shikanov, a professor of biomedical engineering at U-M, in a statement.

Most of the follicles remain inactive and are located in the outer layer of the ovary, called the cortex. Few activate periodically and migrate to the ovary. Only a few follicles— of the growing ones— produce mature eggs that get released into the fallopian tub.
With the ability to direct follicle development and tune ovarian environment, they believe that engineered ovarian tissue could function longer.
What does that mean for patients?
A longer fertility window and a longer period in which their bodies produce hormones.
Those hormones help regulate the menstrual cycle and support muscular, skeletal, sexual, and cardiovascular health.
“We’re not talking about utilizing a surrogate mother, or artificial insemination. The magic we’re working toward is being able to trigger an immature cell into maturity but without knowing which molecules drive that process, we’re blind,” said Jun Z. Li, associate chair of U-M’s Department of Computational Medicine and Bioinformatics and co-corresponding author of the study.
“This was the first time where we could target ovarian follicles and oocytes and perform a transcription analysis. That enables us to see which genes are active. The majority of ovarian follicles, already present at birth, never enter the growing pool and eventually self-destruct,” Shikanov added. This new data allows us to start building our understanding of what makes a good egg—what determines which follicle is going to grow, ovulate, be fertilized, and become a baby.”
Source: Interesting Engineering
Scientists Propose New Method To Detect Consciousness in Infants
Artificial ovary? First atlas of human ovary, a fertility breakthrough